Chapter 20
Submacular Hemorrhage
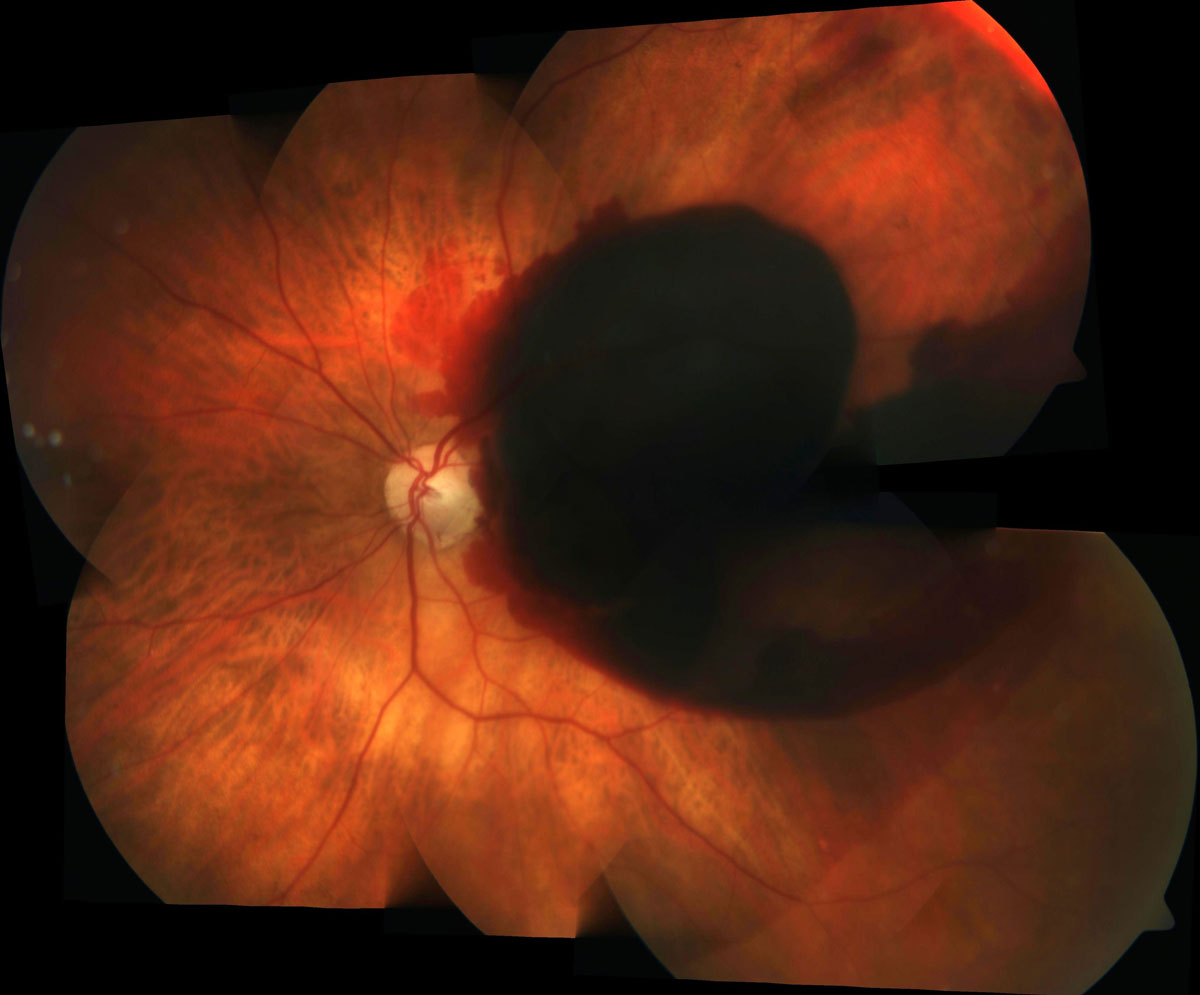
Submacular hemorrhages (SMH) may occur in the following settings (Figure 20.1):
- Choroidal Neovascularization
- Neovascular age-related macular degeneration
- Polypoidal choroidal vasculopathy
- Other causes of Type 1 or Type 2 neovascularization
- Ruptured retinal arterial macroaneurysm
- Traumatic choroidal rupture
The major goals of treatment are evacuating the blood away from macula to avoid potential retinal toxicity and to treat the underlying cause. The treatment options include:
- Intravitreal anti-vascular endothelial growth factor (anti-VEGF)
- Tissue-plasminogen activator (t-PA) - injected intravitreally or into the subretinal space to liquify the clotted blood
- Pneumatic displacement- intravitreal or subretinal injection of air or gas to move the blood away from the macula
- Pars plana vitrectomy (PPV)- to allow access to the subretinal space and increase space for pneumatic displacement The above procedures can be used individually or in combination. Some common strategies with their indications and advantages are outlined in Table 20.1:[1]
Stanescu-Segall D, Balta F, Jackson TL. Submacular hemorrhage in neovascular age-related macular degeneration: A synthesis of the literature. Surv Ophthalmol 2016;61(1):18-32.
Therapy
Indications / Advantages
1. Observation
For small hemorrhages not responsive to anti-VEGF (i.e. non-CNV)
2. Anti-VEGF Monotherapy[2]
Patients who cannot position
Small or chronic hemorrhage
Retains the vitreous reservoir so that the half-life of future intravitreal therapy is not reduced
3. Pneumatic Displacement
± intravitreal t-PA
± intravitreal anti-VEGF
Prompt treatment in office settings
Doesn’t induce a cataract
Retains the vitreous reservoir so that the half-life of future intravitreal therapy is not reduced
4. Vitrectomy + Gas
± subretinal t-PA
± intravitreal anti-VEGF
Larger hemorrhages
Induces a cataract
The half-life of future intravitreal therapy is reduced
Iacono P, Parodi MB, Introini U, et al. Intravitreal ranibizumab for choroidal neovascularization with large submacular hemorrhage in age-related macular degeneration. Retina 2014;34(2):281-287.
Note
A prospective, noncomparative case series demonstrated the benefit of intravitreal t-PA + pneumatic displacement in treating submacular hemorrhages secondary to neovascular age-related macular degeneration.[3] The study showed more favorable results when done within 14 days after the onset of SMH. However, injection of t-PA bears a risk of causing recurrent bleeding and should be done cautiously especially in the acute phase (less than 3 days after the onset of SMH)
Hattenbach LO, Klais C, Koch FH, Gumbel HO. Intravitreous injection of tissue plasminogen activator and gas in the treatment of submacular hemorrhage under various conditions. Ophthalmology 2001;108(8):1485-1492.
The choice of therapy depends on many factors, including:
- Underlying etiology
- In general only CNVs will be responsive to intravitreal anti-VEGF
- Vitrectomy surgery will remove the vitreous reservoir, and may shorten the half-life of future intravitreal anti-VEGF injections
- Duration of hemorrhage
- Acute hemorrhages may be at greater risk of bleeding with t-PA
- Chronic hemorrhages may be less responsive to attempts at displacement
- Amount of hemorrhage
- Thin hemorrhages will resolve faster, and may not require pneumatic displacement
- Level of hemorrhage
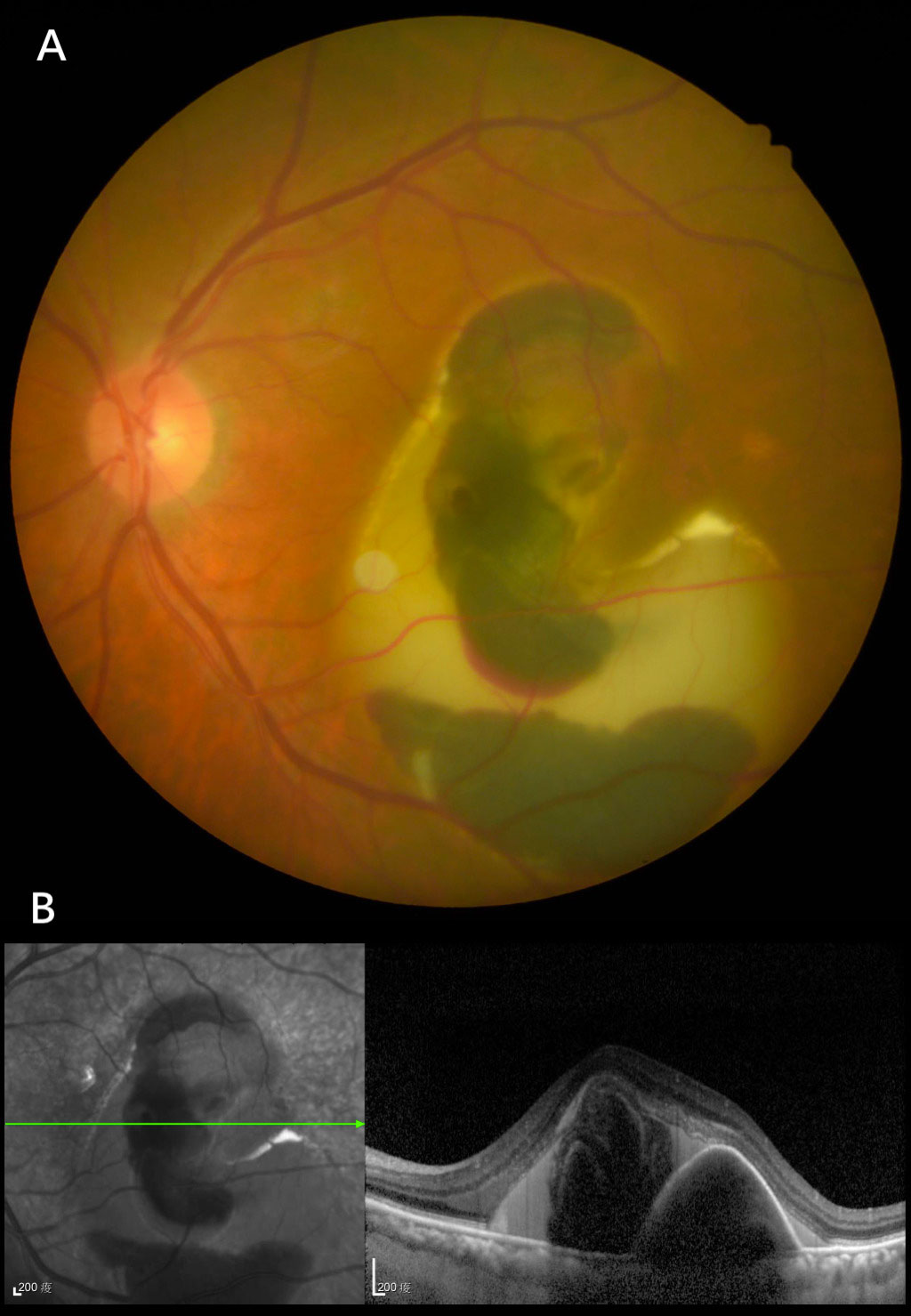
- Subretinal hemorrhages can be displaced, but sub-retinal pigment epithelial hemorrhages cannot be easily displaced. Often there is blood in both levels. In that case one needs to determine if there is significant enough subretinal blood that displacement will be worthwhile. Sub-RPE blood tends to be a darker color than subretinal blood. An OCT is invaluable in determining the level of blood. Fundus autofluorescence can also help differentiate the level of blood. Since lipofuscin in the RPE is hyperautofluorescent, subretinal blood causes blockage of underlying background hyperautofluorescence, whilst sub-RPE blood does not. Eyes with sub-RPE blood may have a more guarded visual prognosis (Figure 20.2)
- Location of hemorrhage in relation to the fovea
- Due to posturing, it is easier to displace hemorrhage inferiorly and temporally than superiorly or nasally. A patient with a predominantly superior macular hemorrhage may not be well served by pneumatic displacement, which risks pushing more blood towards the fovea
- Visual acuity and visual needs
- If the visual acuity is good or the visual needs are low, there will be less enthusiasm for surgical procedures with their associated risks
- Concomitant ocular pathology
- If the affected eye already has poor vision due to other diseases, there will be less enthusiasm for surgical procedures with their associated risk
- Age of the patient
- A very elderly patient with significant co-morbidities may not be suitable for a surgical procedure
- Ability to posture
- Patients who are unable to posture face down may not be suitable for pneumatic displacement
Below is a description a vitrectomy with gas tamponade, subretinal t-PA and anti-VEGF therapy:
1. Opening
The sclerotomy ports and infusion should be set as for a standard vitrectomy. The size of the sclerotomies depend on the available instruments (including subretinal cannula).
All rights reserved. No part of this publication which includes all images and diagrams may be reproduced, distributed, or transmitted in any form or by any means, including photocopying, recording, or other electronic or mechanical methods, without the prior written permission of the authors, except in the case of brief quotations embodied in critical reviews and certain other noncommercial uses permitted by copyright law.
Westmead Eye Manual
This invaluable open-source textbook for eye care professionals summarises the steps ophthalmologists need to perform when examining a patient.